The results indicate that GO/2Pt2Pd catalysts exhibited high activity with high stability toward formic acid and the mixed acids in the biomass product containing FA and AA at a 12.5:87.5 ratio. Figure 21 - XPS peak fitting of (a) Cu 2p doublet at 932 eV (Cu 2p3/2) and.

#PT XPS PEAK FOR PT METAL DOWNLOAD#
In all cases, both noble Pt and Pd in bimetallic catalyst demonstrated that the catalytic ability of them for acid oxidation can be strongly promoted by the appropriate ratio of alloying metal, and the outer layer of Pd and Pt on the electrode affected the oxidation ability of different acid compositions. nanoparticles was investigated in a range of Pt:Cu metal molar ratios varying. Download scientific diagram Deconvolution of relative peaks from XPS spectra at (A) Pt 4f, (B) C 1s and Ru 3d, (C) Ru 3p Pt 0.75 (RuO 2 - MoO 3 ) 0.25 /C, Pt 0.50 (RuO 2 - from publication. can be rationalized by the enhancement of the electrostatic field around Pt nuclei due to the vicinity. In this work, Schottky contacts of Pt and Au on GaN were studied by XPS.
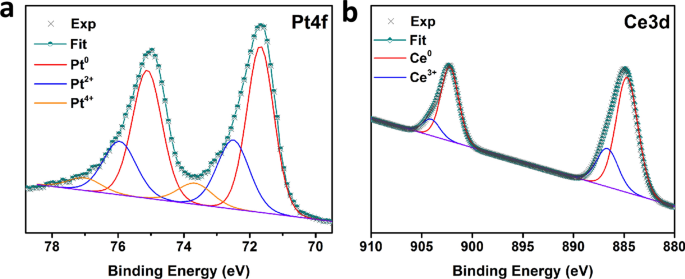
The structure, surface morphology, and elemental composition of the prepared catalysts were also broadly characterized. The Pt 4f 7/2 peak for PtZn ALD /SiO 2 is at 71.7 eV. Graphene oxide (GO) carrying Pt and Pd nanoparticles homogeneously dispersed by electrodeposition was selected as a carbon support for catalyst fabrication for various electrodes to enhance the oxidation of various components of mixed acids (such as formic acid (FA) and acetic acid (AA)). We propose a model, consistent with earlier work on high surface area Pt/TiO2 catalysts, in which the morphology of the Pt on the reduced catalysts is significantly flatter (dominated by (111) terraces) and is slightly modified electronically, as compared to the oxidized sample.This work aims to use mixed acids from biomass production as fuel for low-temperature fuel cells. No evidence for significant differences in the chemical state of the Pt is found by XPS and AES. behavior of the Pt atoms in its vicinity lead- ing to the different catalytic reaction behav- ior characteristic of bimetallic catalysts as opposed to monometallic Pt/AI203 cata- lysts. Platinum is named for the Spanish word platina, meaning little. It is also used to make surgical tools, liquid crystal displays, and electrical resistance wires. Because of its extreme durability and resistance to tarnishing, platinum is widely used to make jewelry.
The state of Pt is confirmed by using XPS. Platinum is very rare, and is naturally found as the uncombined metal. In particular, at a given Pt coverage there is less chemisorption of both CO and H2 on the reduced, as compared to the oxidized, TiO2 and the desorption spectra peak at lower temperatures. The weak and broad peaks appear at 2 39, indicating Pt metal exists on GNS. As a function of Pt coverage, the development of bulk Pt chemisorption characteristics occurs at significantly lower coverages on the oxidized substrate. Both fully oxidized (Ti(4+)) and partially reduced (significant amounts of Ti(3+)) surfaces were studied. Thin films of Pt, vapor deposited on an oxidized Ti foil, have been utilized as models of high surface area Pt/TiO2 catalysts which show strong metal-support interactions (SMSI).
